Biofuel industry news
Published over 4 years ago. See the latest and most current information on Biofuel industry news.
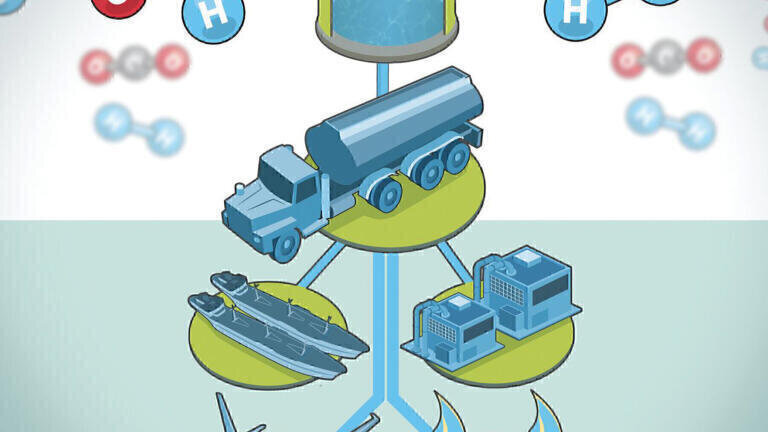
Carbon emissions have plagued our planet to a point where a change is required to reverse any adverse effects on the environment. Global temperatures are rising at an alarming rate of over 20C a year. The impending climate disaster is the reason why the search for sustainable fuels has become a focal point in the fuel industry. The design and development of innovative electricity-generated fuels (e-fuels) have led to promising results of a product fuel that has a net-zero carbon footprint. The basis of e-fuel production is to convert electricity to hydrogen gas (H2) and use captured carbon, in the form of carbon dioxide gas (CO2), to both react together and form an e-fuel. The reaction between the CO2 and H2 gases is most commonly utilized and transformed into a product of synthetic hydrocarbon gases like methane (CH4) or butane, or in liquid form alcohol fuels like methanol (CH3OH) [1]. Figure 1 presents a visual of the processes necessary to produce these synthetic e-fuels.
The process of the creation of a methanol-based fuel will be discussed as a potential replacement for the traditional petroleum-based fuel. In addition, the benefits of e-fuels will be highlighted to further prove why this swap is necessary. The future scope is bright as the use of e-fuels can steer the way for a cleaner and more renewable future.
E-fuels have been revolutionary in the continuous search for a sustainable clean fuel source. Experts have experimented with many different processes in an attempt to find the most efficient process, financially and energy-wise, to output the best product. The use of renewable electricity as the dominant source of energy and the main raw material used in the production of water and carbon dioxide is a commonality in the majority of e-fuel processes. Each different e-fuel, formulated from different processes, has its own unique features that can be specifically helpful for certain applications. Many different reactions have been experimented with to produce e-fuels. Some of the resulting e-fuels from various methods used include hydrogen, methane, methanol, dimethyl ether (DME), Fischer-Tropsch synthetic fuel products, oxymethylene ethers (OME), and mixtures of dimethylcarbonate and methylformiate [3]. While many of these methods have proven to be successful, some have exceeded expectations and the resulting e-fuel product is comparable to the performance and cost of traditional petroleum-based fuels. Focusing on the synthesis of methanol through a sequence of reactions is explored throughout this paper.
In the earliest stage of formulating an e-fuel, hydrogen gas must be produced. The common and reliable method of hydro electrolysis is a clean and effective way of obtaining hydrogen gas. The reaction takes place in an electrolyzer which consists of an anode and a cathode separated by an electrolyte. When water is introduced to the system, it reacts at the anode to form oxygen and positively charged hydrogen ions. The electrons flow through an external circuit and the hydrogen ions selectively move across the polymer electrolyte membrane to the cathode where the hydrogen ions combine with electrons from the external circuit to form hydrogen gas [4]. The visual representation of this process and the reaction equations can be viewed in Figure 2.
Now that hydrogen gas has been obtained, it must be reacted with a carbon compound to be used as a fuel base. To further improve environmental conditions, carbon dioxide is captured and reused in the e-fuel production process as the required carbon compound. Excess CO2 can be obtained from injected CO2 geological storage locations including coal beds, deep saline formations, shale formations, or oil and gas reservoirs [5] but, to further improve environmental conditions the excess CO2 from processes such as recycling or biomass gasification is captured by an energy storage technique/system and used to formulate an e-fuel [6]. One of the most prevalent carbon capture methods utilized in the production of an e-fuel is physical separation of the CO2. Physical adsorption or absorption is the principal of this process. This can be done by a solid surface, such as metallic oxides, adsorbing the CO2 gas or via a liquid solvent (typically Selexol or Rectisol) to absorb the CO2 gas [7]. The CO2 gaseous form can be released at a later time by increasing temperature or pressure from the adsorbent. In addition to physical separation, a newer carbon capture technique performed to be used in the production of synthetic e-fuels is direct air capture (DAC). The DAC process starts with an air contactor where a large fan pulls air into the system. Once the air has entered the system it’ll pass over thin plastic surfaces that have a non-toxic solution of potassium hydroxide flowing over them. The CO2 molecules chemically bind with the solution, removing them from the air and trapping them in the liquid solution as a carbonate salt [8]. To release a concentrated CO2 gas, to be further utilized, the gas must first be separated from the salt out from the solution. The subsequent reaction, in a pellet reactor, yields a precipitate that then gets passed through a calciner. The precipitated pellets are heated in the calciner (renewable sources such as hydroelectric, wind, or solar energy can be used to power the calciner) to obtain our desired CO2 in pure gas form. After the calciner reaction concludes the pellets are recycled back into the system to reproduce the original capture chemical [8]. The DAC process is continued to be researched as there are some concerns with the amounts of energy it takes to heat the reaction that separates the CO2 gas. It is critical to note that by utilizing either of these methods maintains net-zero carbon emissions.
The particular alternative fuel source of methanol has been studied intensively and determined to be a reliable fuel alternative. The captured carbon dioxide is processed to methanol through the process of direct carbon dioxide hydrogenation. Direct carbon dioxide hydrogenation has been shown to carry out this process with promising results due to the reduced heat necessary to perform the reaction as opposed to the process of carbon dioxide hydrogenation via a reverse-water-gas-shift reaction. This shift reaction, from CO2 to CO, has been utilized due to being able to use the desirable copper catalyst. Although, a different metal catalyst would have to be used for the reaction between CO2 and H2 gases because this reaction produces water, which causes the copper catalyst to shut down. In Figure 3, the reaction equations for each process are shown.
Figure 3. Hydrogenation reaction equations of CO2/CO and H2 [9]
A study from Nankai University concludes that the precursor of CO2 can yield a methanol product with the use of a Ruthenium (Ru) catalyst. The metric used to measure the effectiveness of a catalyst is the turnover number. The turnover number is defined as the number of substrate molecules transformed per minute by a single enzyme molecule [10]. Catalyst Ru3(CO)12 was experimentally tested and was determined to have a turnover number of 94.5 with the potential of achieving a turnover number of up to 221 in the presence of an acidic additive [9]. The main concern of the use of the Ru catalyst is the fact that the system would often suffer from harsh reaction conditions such as high temperatures and pressures along with the presence of co-products. In contrast, a Nickel (Ni) catalyst has also been explored due to how inexpensive Ni catalysts tend to be along with the lower reaction temperatures observed during methanol synthesis and CO2 elimination [9]. The Ni catalyst was shown to produce a high yield of CH3OBcat which can be used as a methanol precursor [9]. Another plus to the reaction with the Ni catalyst is that the reaction is reversible. So, why are we evaluating methanol as opposed to other possible e-fuels?
As seen in Figure 4, methanol-based e-fuels are among the lowest in energy required to produce the fuel. The storage and transport of methanol e-fuels is also a massive advantage. If there was to be a methanol spill in the ocean the methanol would dissolve within 24 to 48 hours with virtually no negative environmental impacts [12]. In addition, methanol has a density comparable to a typical petroleum-based fuel which means no additional pressure is required in the storage of the methanol e-fuels unlike in the storage of hydrogen-based e-fuels. With petroleum-based fuels expected to be maintained as the dominant fuel source for the foreseeable future, methanol fuels have been experimentally determined to be an effective carbon neutral additive to diesel fuels to reduce the CO2 emissions of the current most important popular backup fuel in the power generation sector [11]. Ultimately, the conversion from excess CO2 to a reliable e-fuel has been successfully achieved. As the combination of these technologies is still relatively new, it’s just a matter of time until these processes become sustainable and the potential rise of a new dominant fuel source is seen.
With the rise of every new fuel source, the pros and cons must be weighed. The efficiency and effectiveness of the resulting e-fuel along with the environmental benefits has made e-fuels a viable option for the future. The first major benefit of e-fuels to be discussed is how e-fuels are environmentally and climate-friendly fuels. E-fuels produced using renewable electricity do not produce any carbon emissions in the process. Since renewable electricity is preferred, this would provide a platform for growth in many different types of renewable energy sources such as solar and wind power. The potential of e-fuels to be utilized in daily processes, such as in an engine of a vehicle, allows for the incorporation of sustainability into existing infrastructure in both the transportation and heating industry sectors [13].
In order to determine how environmentally friendly e-fuels are, we must talk about the source from which e-fuels are made. This source is renewable energy. A study was done comparing the CO2-eq emissions from wind energy, fossil fuels, and a German electricity mix for electrolysis. Both wind energy and the German electricity mix can be used as sources for the e-fuels. Figure A1 shows that wind energy as the source produces significantly fewer carbon emissions than both fossil fuels and the German electricity mix. Therefore, it is important to use renewable sources of energy when designing e-fuels [14]. Figure A1 also breaks down where most of the carbon dioxide emissions are coming from. The electrolysis is what makes up most of the CO2-eq emissions produced by German electricity mix while there are no CO2-eq emissions produced by electrolysis using wind energy.
This environmentally friendly trait that e-fuels have is further backed up by the Greenhouse Gas Emissions (GHG) that are produced by the fuel. Figure A2 illustrates the German Electricity Mix being substituted by renewable energy in a stepwise manner. As depicted, the use of wind energy for electrolysis has a major reduction in CO2-eq emissions by about 85%. As additional renewable energy sources were substituted in, the fewer CO2-eq emissions were produced. We can conclude that it is essential that the electrolysis is powered by renewable electricity for e-fuels to achieve low CO2-eq emissions [14].
Another essential benefit of e-fuels is how economically favorable e-fuels are to the fuel industry. In the analysis of economic benefits, it is important to first look at the production costs currently for e-fuels. The biggest contributing factor to the cost of e-fuels comes from the production of hydrogen. The hydrogen can come from both renewable and non-renewable sources. However, e-fuels require hydrogen from renewable processes in order to maintain net-zero carbon emissions produced. The most used of these processes is electrolysis. There are multiple types of electrolyzers and, thus, electrolysis processes that are performed through these different types of electrolyzers which ultimately result in different compositions of an e-fuel that each has its own associated cost. This is depicted in Figure A3. The low and high values in Figure A3 depict the most optimistic and pessimistic values in the literature, these values are used for the efficiencies and costs of electrolyzers and fuel synthesis, respectively and the average data is used as the base [2].
We can conclude from Figure A3 that e-fuel production costs can be kept as low as less than 100 €/MWh which is comparable to typical petroleum-based fuels. It is also observed that over the five years of this study the production costs of e-fuels have been significantly lowered, some by over 50%.
The most unique benefit of e-fuels is the high energy density found within them. High energy density is related to the relative easiness of the ability to store fuels and thus helps maintain a low storage cost [15]. This can be compared to other types of fuels as well. A study from 2021 shows a comparison of e-fuels being used in fuel cells and comparing them to alcohol fuel cells. The results of e-fuels being used as the anode are shown in Figure A4. As can be seen from Figure A4, the cell achieves an energy efficiency of 41.8% at 200 mA/cm2 [16] which is much higher efficiency than conventional alcohol fuel cells. Conventional alcohol fuel cells have about a 7% energy efficiency [17].
E-fuels have numerous benefits to be considered. These advantages are reasons why e-fuels are looked at as a possible solution to reducing carbon emissions in the future. Analyzing how e-fuels are currently being used can get us an idea of how they may advance in the future.
Projects to commercialize e-fuels are already underway. In specific, the company Carbon Recycling International (CRI) has been a pioneer in the development of CO2 to methanol plants. Their current projects include the Shuni CO2-to-methanol plant, George Olah renewable methanol plant, and MEFCO2. These facilities have received millions of dollars in funding and for good reason as the George Olah plant has the capacity to recycle 5500 tons of carbon dioxide emissions [18] and the yearly production rate reaches 5 million liters of methanol fuel, which amounts to about 2.5% of the Icelandic fuel consumption [3]. Another company making a major impact in the e-fuels industry is Velocys’. Velocys’ is focused on the Fischer-Tropsch (FT) process in creating an e-fuel meant for jets. The company’s most recent accomplishment, in 2021, was the second successful woody biomass to jet fuel demonstration in Japan. The flight successfully traveled from Tokyo to Sapporo on June 17th, 2021. The fuel assembled for the flight was confirmed to fully conform to ASTM D7566 and the full amount of the FT synthesis technology neat biojet fuel produced since then (2,366L) were also confirmed its conformity [19]. As more companies embark on their adventures into experimenting with e-fuels we should see a sharp rise in production and usage of e-fuels.
The future of e-fuels is bright. It is imperative that explorations on developing more efficient and cost-effective ways to produce H2 from water electrolysis and implementation of renewable energy for CO2 hydrogenation are researched [20]. New technologies for renewable energy and H2 production will only continue to grow. Many nations will revert to renewable energy rather than fossil fuels in the future. One such country is Denmark which aims to become fossil fuel independent within its transport sector. An analysis of an anticipated 2050 Danish Energy System report shows that e-fuels that use biomass would thrive within this system. It’s stated that e-biofuels would make up 43% of the transport sector of the base model utilized. In all of the scenarios, the total production of e-biofuels and e-fuels is at least 26% [21]. Germany has also mapped out a scheme to reduce current CO2 emissions by an additional 50% to total a 95% change from 1990 by 2050. Their plan includes 80% of the nationwide electricity supply be supplied by renewable energies by 2050 [22]. At the 2021 United Nations Climate Change Conference (COP26) it was recognized that member countries must pledge to further cut emissions of carbon dioxide to a rate where there is a maximum of 1.5℃ increase in global temperatures per year. If countries were to continue with current regulations, global temperatures can increase to a concerning rate of 2.5℃ per year. However, in 2020, global CO2 emissions from fossil fuels fell 7%, the largest annual drop ever observed [23]. An e-fuel’s ability to obtain net-zero carbon emissions can contribute to further lowering emissions if usage is increased. With continued research and development of sustainable e-fuel methods, we can continue lowering emissions and all in all, prevent the imminent climate disaster.
Dr. Raj Shah is a Director at Koehler Instrument Company in New York, where he has worked for the last 27 years. He is an elected Fellow by his peers at IChemE, CMI, STLE, AIC, NLGI, INSTMC, Institute of Physics, The Energy Institute and The Royal Society of Chemistry. An ASTM Eagle award recipient, Dr. Shah recently coedited the bestseller, “Fuels and Lubricants handbook”, details of which are available at https://bit.ly/3u2e6GY
He earned his doctorate in Chemical Engineering from The Pennsylvania State University and is a Fellow from The Chartered Management Institute, London. Dr. Shah is also a Chartered Scientist with the Science Council, a Chartered Petroleum Engineer with the Energy Institute and a Chartered Engineer with the Engineering council, UK. Dr. Shah was recently granted the honorific of “Eminent engineer” with Tau beta Pi, the largest engineering society in the USA. He is on the Advisory board of directors at Farmingdale university (Mechanical Technology), Auburn Univ (Tribology) and Stony Brook University (Chemical engineering/ Material Science and engineering). An Adjunct Professor at the State University of New York, Stony Brook, in the Department of Material Science and Chemical engineering, Raj also has over 475 publications and has been active in the petroleum industry for over 3 decades.
More information on Raj can be found at https://bit.ly/3sayVgT
Anthony Schevon and Hugo Ramos are Chemical Engineering students from SUNY, Stony Brook University, where Dr. Shah is the chair of the external advisory board of directors. They are also a part of a growing internship program at Koehler Instrument Company in Holtsville, NY that helps students explore opportunities in the field of Alternative energy.
[1] H. Stančin, H. Mikulčić, X. Wang, N. Duić, A review on alternative fuels in future energy system, Renewable and Sustainable Energy Reviews, Volume 128, 2020, 109927, ISSN 1364-0321, https://doi.org/10.1016/j.rser.2020.109927.
[2] Brynolf, Selma, et al. “Electrofuels for the Transport Sector: A Review of Production Costs.” Renewable & Sustainable Energy Reviews, vol. 81, no. 2, Elsevier Ltd, 2018, pp. 1887–905, https://doi.org/10.1016/j.rser.2017.05.288.
[3] Ausfelder, F., & Wagemann, K. (2020). Power‐to‐fuels: E‐fuels as an important option for a climate‐friendly mobility of the future. Chemie Ingenieur Technik, 92(1-2), 21–30. https://doi.org/10.1002/cite.201900180
[4] Hydrogen production: Electrolysis. Energy.gov. (n.d.). Retrieved March 1, 2022, from https://www.energy.gov/eere/fuelcells/hydrogen-production-electrolysis
[5] Carbon capture. Center for Climate and Energy Solutions. (2021, May 27). Retrieved from https://www.c2es.org/content/carbon-capture/
[6] B.V. Mathiesen, H. Lund, D. Connolly, H. Wenzel, P.A. Østergaard, B. Möller, S. Nielsen, I. Ridjan, P. Karnøe, K. Sperling, F.K. Hvelplund, Smart Energy Systems for coherent 100% renewable energy and transport solutions, Applied Energy, Volume 145, 2015, Pages 139-154, ISSN 0306-2619, https://doi.org/10.1016/j.apenergy.2015.01.075.
[7] IEA (2021), About CCUS, IEA, Paris https://www.iea.org/reports/about-ccus
[8] Direct Air Capture Technology. Carbon Engineering. (2021, December 13). Retrieved March 1, 2022, from https://carbonengineering.com/our-technology/
[9] Li, Y.-N., Ma, R., He, L.-N., & Diao, Z.-F. (2014). Homogeneous hydrogenation of carbon dioxide to methanol. Catal. Sci. Technol., 4(6), 1498–1512. https://doi.org/10.1039/c3cy00564j
[10] Anthony Y.H. Lu, Gerald T. Miwa, Susan B. West, ON THE STIMULATORY ROLE OF PHOSPHATIDYLCHOLINE IN A CYTOCHROME P-450-CONTAINING RECONSTITUTED HYDROXYLATION SYSTEM, Editor(s): Minor J. Coon, Allan H. Conney, Ronald W. Estabrook, Harry V. Gelboin, James R. Gillette, Peter J. O’Brien, Microsomes, Drug Oxidations and Chemical Carcinogenesis, Academic Press, 1980, Pages 59-66, ISBN 9780121877019, https://doi.org/10.1016/B978-0-12-187701-9.50011-5.
[11] Krol, T., & Lenz, C. (2020). Can e-fuels close the renewables power gap? A review. VGB PowerTech.
[12] Sporrer, A. (2021, October 7). E-methanol: Missing piece to shipping’s decarbonization puzzle? FreightWaves. Retrieved from https://www.freightwaves.com/news/e-methanol-missing-piece-to-shippings-decarbonization-puzzle
[13] Efuels – sustainable in two ways. eFuel Alliance. (n.d.). Retrieved March 1, 2022, from https://www.efuel-alliance.eu/efuels
[14] Laura E. Hombach, Larissa Doré, Katrin Heidgen, Heiko Maas, Timothy J. Wallington, Grit Walther, Economic and environmental assessment of current (2015) and future (2030) use of E-fuels in light-duty vehicles in Germany, Journal of Cleaner Production, Volume 207, 2019, Pages 153-162, ISSN 0959-6526, https://doi.org/10.1016/j.jclepro.2018.09.261.
[15] Bothe, D. (2019, July 10). E-fuels from Renewable Electricity Key for a successful energy turnaround. Frontier Economics. Retrieved from https://www.frontier-economics.com/uk/en/news-and-articles/news/news-article-i6477-e-fuels-from-renewable-electricity-key-for-a-successful-energy-turnaround/#
[16] Shi, Xingyi, et al. “Performance Characteristics of a Liquid e-Fuel Cell.” Applied Energy, vol. 297, Elsevier Ltd, 2021, p. 117145–, https://doi.org/10.1016/j.apenergy.2021.117145.
[17] Wang, Lianqin, et al. “Energy Efficiency of Alkaline Direct Ethanol Fuel Cells Employing Nanostructured Palladium Electrocatalysts.” ChemCatChem, vol. 7, no. 14, WILEY-VCH Verlag, 2015, pp. 2214–21, https://doi.org/10.1002/cctc.201500189.
[18] Projects: Emissions-to-liquids technology. CRI - Carbon Recycling International. (n.d.). Retrieved from https://www.carbonrecycling.is/projects
[19] Achieved the flight using sustainable aviation fuel (SAF) made from woody biomass. Toyo Engineering Corporation. (2021, June 18). Retrieved March 1, 2022, from https://www.toyo-eng.com/jp/en/company/news/?n=584
[20] Xiao Jiang, Xiaowa Nie, Xinwen Guo, Chunshan Song, and Jingguang G. Chen, Recent Advances in Carbon Dioxide Hydrogenation to Methanol via Heterogeneous Catalysis, Chemical Reviews 2020 120 (15), 7984-8034 DOI: 10.1021/acs.chemrev.9b00723
[21] Lester, Mason Scott, et al. “Analysis on Electrofuels in Future Energy Systems: A 2050 Case Study.” Energy (Oxford), vol. 199, Elsevier Ltd, 2020, p. 117408–, https://doi.org/10.1016/j.energy.2020.117408.
[22] Steffen Schemme, Remzi Can Samsun, Ralf Peters, Detlef Stolten, Power-to-fuel as a key to sustainable transport systems – An analysis of diesel fuels produced from CO2 and renewable electricity, Fuel, Volume 205, 2017, Pages 198-221, ISSN 0016-2361, https://doi.org/10.1016/j.fuel.2017.05.061.
[23] CB Insights. (2021, April 7). Direct air capture explained: The Buzzy New Carbon Reduction Tech gaining exec attention. CB Insights Research. Retrieved March 1, 2022, from https://www.cbinsights.com/research/direct-air-capture-corporate-carbon-reduction/
PIN 27.2 Apr/May 2026


